“In metric, one milliliter of water occupies one cubic centimeter, weighs one gram, and requires one calorie1 of energy to heat up by one degree centigrade—which is 1 percent of the difference between its freezing point and its boiling point. An amount of hydrogen weighing the same amount has exactly one mole of atoms in it. Whereas in the American system, the answer to ‘How much energy does it take to boil a room-temperature gallon of water?’ is ‘Go fuck yourself,’ because you can’t directly relate any of those quantities.” Wild Thing by Josh Bazell.
I have not read Wild Thing, and I do not have any idea about who Josh Bazell is, but I love this quote. It brilliantly summarises how superior the International System of Units (i.e., the modern form of the metric system) is to the system of imperial units, as shown in the following illustration:
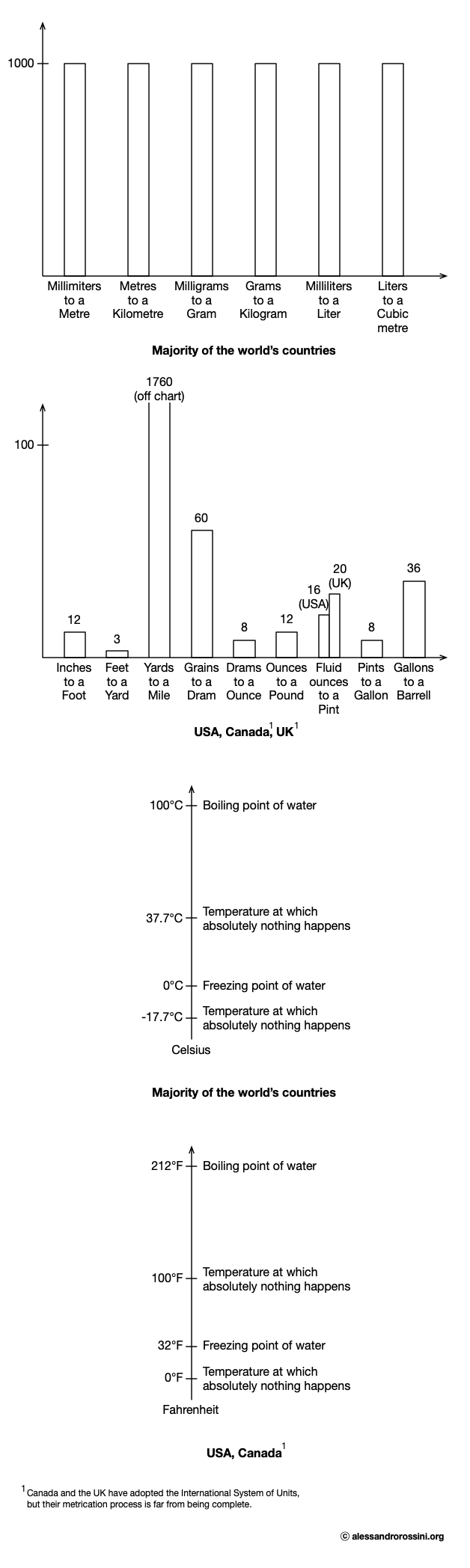
Yet, nowadays…
The USA is the only industrialised country in the world that still uses a system of units developed from English units (i.e., the United States customary system), as well as the only industrialised country in the world that has not adopted the International System of Units2(see Metrication in the USA).
Canada and the UK have adopted the International System of Units, but their metrication process is far from being complete (see Metrication in the UK and Canada).
Despite the fact only a minority of countries have not adopted the International System of Units, the majority of countries are still contaminated by the system of imperial units. Think about it… Even in continental Europe, where the metric system comes from and where the imperial units are nowhere in the education curriculum, screens are measured in inches, aircrafts’ altitude is measured in feet, watercrafts’ speed in measured in knots, etc.
The problem is that, to anybody who grew up with the International System of Units, an advertisement of a 55” TV screen tells very little about the actual size; a captain announcing that the plane is cruising at 30,000 feet tells very little about the actual altitude; a speedometer showing that the boat is cruising at 20 knots tells very little about the actual speed.
This is ridiculous and has got to stop. Governments of all countries adopting the International System of Units have to enforce that this system is used in absolutely every application and that any trace of imperial units is consigned to history.
If you are a man or woman of science, or if you simply have common sense, please share this post on every social network, and tag your tweets and instagrams with the tag #banimperialunits.
Here are some examples of tweets that you can send to your politicians.
Inch is not an official unit in Norway. Why are screens measured in inches and not centimetres? #banimperialunits
Foot is not an official unit in Italy. Why does the aviation industry use feet and not metres? #banimperialunits
Knot is not an official unit in Spain. Why does the ship industry use feet and not metres? #banimperialunits
Together, we can put an end to imperial units.
Footnotes
- One may argue that calorie is not a unit of the metric system. In the International System of Units, which is an evolution of the metre-kilogram-second (MKS) system of units, which in turn is a variant of the metric system, both mechanical and thermal energies are measured in Joule. However, in the centimetre-gram-second (CGS) system of units, which is another variant of the metric system, the thermal energy is measured in Calories. Therefore, I would still consider Calorie as one of the units of the metric system, although not of the International System of Units. Besides, I do not think Josh Bazell aimed at being scientifically rigorous, so I would excuse him for not clarifying which variant of the metric system he refers to. ↩
- The two other countries that do not adopt the International System of Units are Myanmar and Liberia, while the four other countries that adopt Fahrenheit for everyday applications are Bahamas, Belize, the Cayman Islands, and Palau. ↩